Resources
VigiLanz Recognized for High Customer Retention, Innovation in KLAS 2019 Infection Control Report
MINNEAPOLIS – March 14, 2019 – VigiLanz, a clinical surveillance company, has been featured in the Infection Control 2019 Report from KLAS Research, released today. The report aggregates and analyzes interviews from thousands of healthcare professionals about the products and services their organizations use.
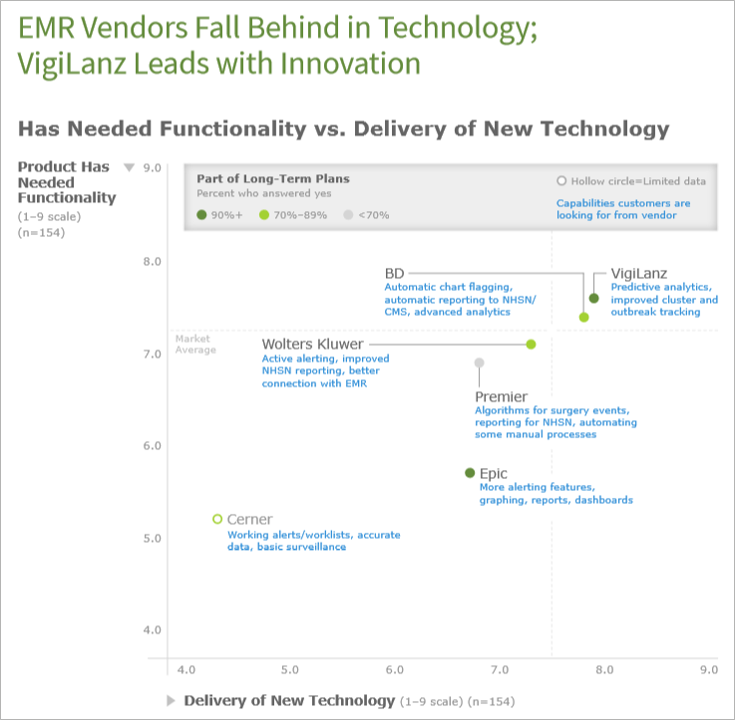
In the report, KLAS characterizes VigiLanz as a market leader for its high customer retention and innovative technology additions, such as active alerting, in its Dynamic Infection Control Monitor product. VigiLanz is also recognized for providing advanced automation and predictive analytics, two capabilities KLAS identified as highly desired by the market.
“Before we had [VigiLanz’s] Dynamic Infection Control Monitor, I did everything with manual data extraction,” said one anonymized user in the report. “I would have to run about 12 different reports to even get a list of patients to perform surveillance on, and I would need another six reports to get all the communicable disease reporting. [The] Dynamic Infection Control Monitor has made my job so much easier. It is a lot more efficient. I can easily get my patient list and go through it.”
Another anonymized user said, “We consider VigiLanz to be the leader in surveillance because of the flexibility of what we can do with the surveillance.”
VigiLanz’s Dynamic Infection Control Monitor is a seamless, real-time automated monitoring system that works with EMR systems. Its automatic exception-based alert system continuously surveils all patient records to alert decision makers in real time of potential infections, drug-bug mismatches, MDROs, isolation candidates, organism clusters, conditions of concern, and reportable infections. The surveillance is based on a sophisticated set of customizable rule engines.
“We understand how critical an infection prevention product is for patient outcomes and hospitals’ bottom lines, and we’re dedicated to the ongoing innovation that ensures we continue delivering what patients and the market needs,” said David Goldsteen, MD, CEO of VigiLanz. “In general, healthcare executives say infection prevention is one of the top five uses of clinical surveillance technology,[1] and it has rapidly become a critical component of quality patient care.”
The other top uses of clinical surveillance technology, according to healthcare executives, are:
1) Identifying adverse drug events
2) Promoting antimicrobial stewardship
3) Patient safety alerting
4) Managing readmissions
In addition to being a leader in this report, KLAS recognized VigiLanz in the 2019 Best in KLAS report, earning 2019 Category Leader designations for Antimicrobial Stewardship, Infection Control and Monitoring, and Pharmacy Surveillance.
About KLAS
KLAS is a research firm on a global mission to improve healthcare delivery by enabling providers and payers to be heard and counted. Working with thousands of healthcare professionals, KLAS gathers insights on software, services and medical equipment to deliver timely reports, trending data and statistical overviews. KLAS data is accurate, honest and impartial. The research directly represents the voice of healthcare professionals and acts as a catalyst for improving vendor performance. To learn more about KLAS and the insights we provide, visit www.KLASresearch.com.
About VigiLanz
Founded in 2001, VigiLanz (www.vigilanzcorp.com) is a privately held, rapidly growing provider of SaaS-based clinical surveillance solutions. The firm is focused on aggregating disparate EMR transactional workflow and documentation data across health systems to identify real-time clinical issues that avoid or minimize harm, optimize clinical outcomes and support preventive care. VigiLanz supports a large and growing community of hospital CMOs, CMIOs, CIOs, quality teams, infectious disease and control specialists, pharmacists, and other clinicians dedicated to real-time inpatient and outpatient care.
[1] Market Report: Clinical Surveillance: The Next Step in Value-Based Care. Feb. 2019. VigiLanz.
Unlocking Patient Safety: The Power of Autodetection in Healthcare
The key to preventing safety events and reducing harm is not about what you know is happening, but what you don’t know. Hear from Dr. Chris Emerson on how autodetection can help healthcare leaders gain visibility into the true state of patient safety inside their institutions.
Enhancing Patient Safety with Automated Event Detection: A Hospital’s Guide
Discover the benefits of autodetection, why every hospital should deploy it, and how one innovative health system is using it to enhance patient safety.
